PharmAI Discovery’s DiscoveryEngine achieves industry-leading performance in early drug discovery, with 30% hit rates in vitro and 16% in vivo, validated through comprehensive biological testing.
Powered by interpretable AI, deep learning, and structure-based modeling, DiscoveryEngine explores an 80-billion-dimensional chemical-biological search space and screens against over 95% of the human proteome. This enables rapid identification of potent, selective compounds, including those targeting complex challenges like protein–protein interactions.
The platform’s built-in in vitro and in vivo validation loop ensures predictions lead to real biological activity, while its off-target profiling capabilities de-risk candidates early in the pipeline. With a track record of 40+ successful projects, DiscoveryEngine transforms discovery timelines and success rates, from digital screening to preclinical proof.
Years Of Research
Our disruptive software technology identifies drug candidates based on the geometry of protein binding sites and their drug-target interactions. This method paves new ways to explore the drug-target space at a ground-breaking precision.

Case study: binders to cGMPdependent 3',5'-cyclic phosphodiesterase (PDE2)
PharmAI’s DiscoveryEngine scored 125 compounds from a library of 2M compounds, spread across the chemical space. After in vitro validation, seven compounds showed binding to PDE2 (at a hit rate of almost 6%) of which three showed affinities in the lower micromolar range.

Our Artificial Intelligence (AI) initiative opens the door to new compound classes. We use classical AI methods with the combination of Deep Learning to predict binding affinity and to improve the quality of our algorithm’s output.
Our aim is make the discovery of drugs cheaper and quicker while keeping it at a high precision. In addition, we contineously improve our algorithms by combining classical and semi-supervised deep-learning in a smart manner.
Case study: Prediction of MAPK14 Off-Targets
Using PharmAI's DiscoveryEngine, 13 off-targets were identified and further validated in the laboratory tests in collaboration with 2bind GmbH.
Of the 13 tested proteins, binding was observed for six which corresponds to an extraordinarily high hit rate of 46 percent.
in collaboration with 2bind GmbH

In Silico Driven Prediction of MAPK14 Off-Targets Reveals Unrelated Proteins with High Accuracy
We invested 500 CPU years of pre-computation to ensure a fast delivery of your results just in a few days. Our efficient algorithms make use of this data resource and run order of magnitudes faster than traditional virtual screening.
There are many reasons for choosing us as your partner in your drug discovery journey.
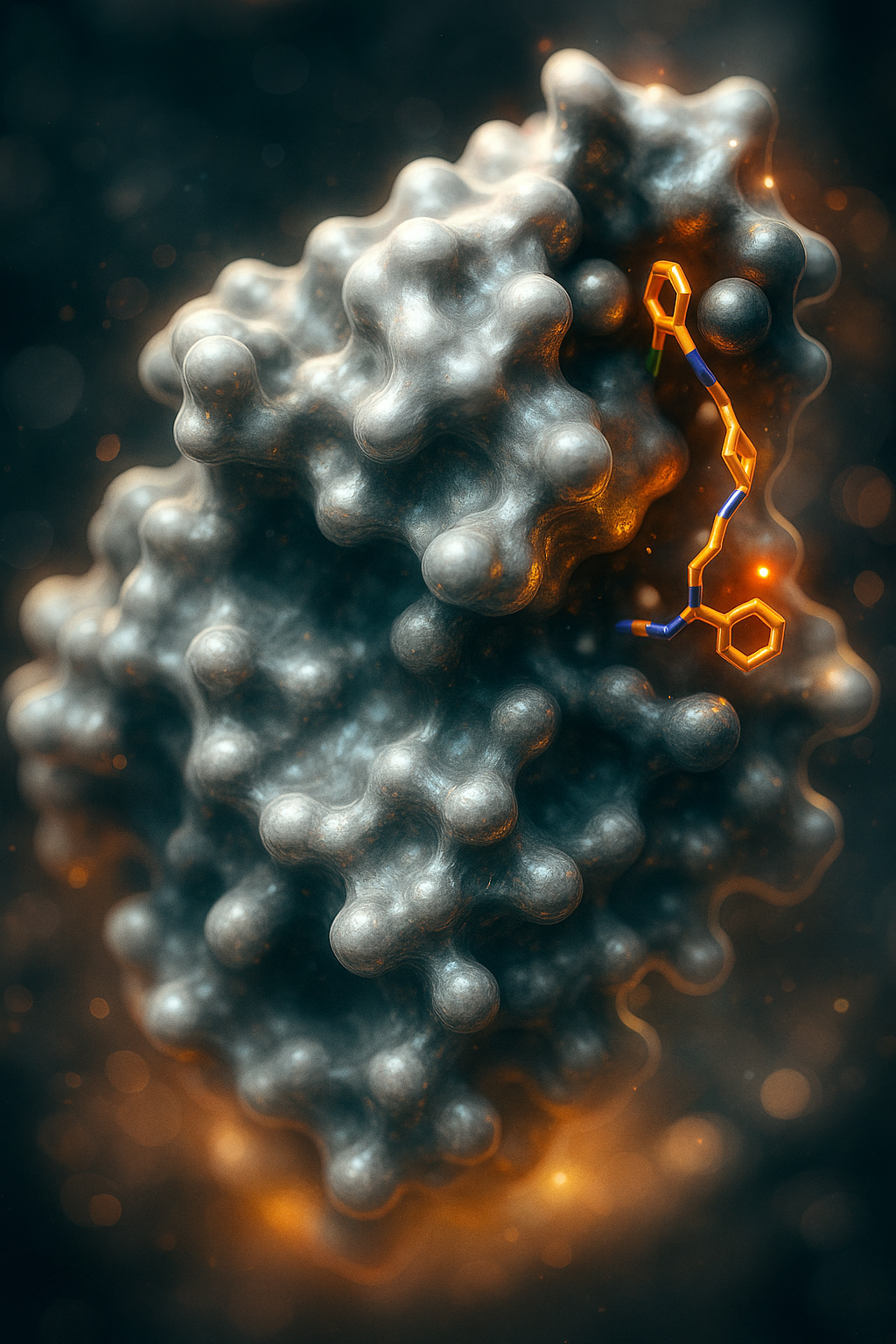
PharmAI's DiscoveryEngine easily explores new chemical scaffolds for your target

Our fingerprinting technology guarantees an accurate representation of your target of interest.

We have pre-invested over 500 CPU years of computation time to deliver your results within a few days.
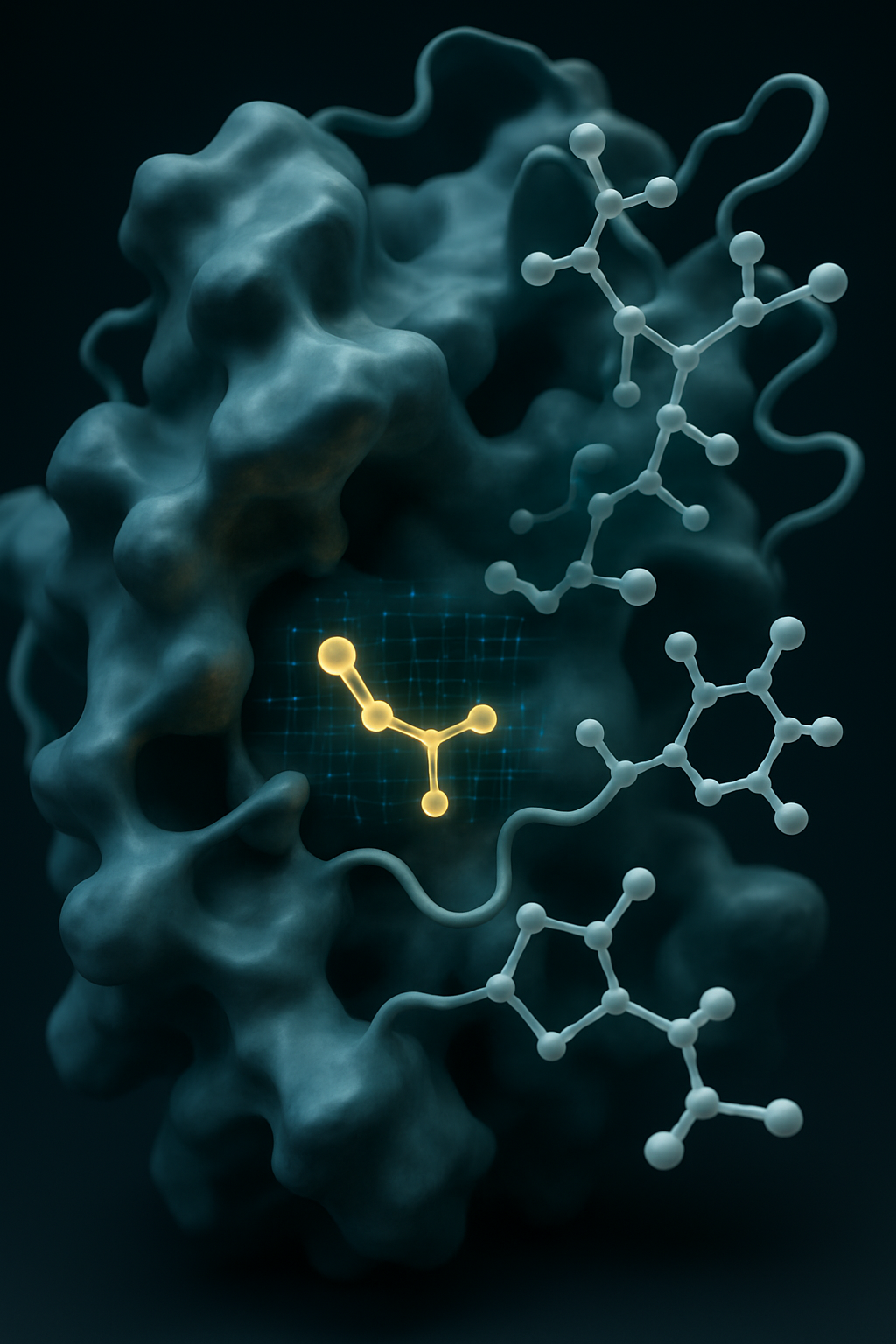
We cover over 80 billion compounds from multiple vendors and can even use your custom library.
PLIP, a framework and a web service for a fully automated characterization of non-covalent interactions between proteins and ligands in 3D structures is now available for the community under Apache License at https://github.com/pharmai/plip.
You can also read about and follow the latest developments of PLIP in our blog.